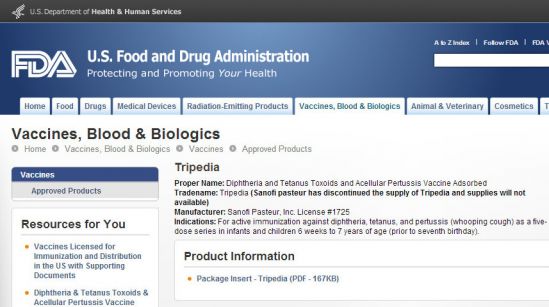
Tripedia
Proper Name: Diphtheria and Tetanus Toxoids and Acellular Pertussis Vaccine Adsorbed
Tradename: Tripedia (Sanofi pasteur has discontinued the supply of Tripedia and supplies will not available)
Manufacturer: Sanofi Pasteur, Inc. License #1725
Indications: For active immunization against diphtheria, tetanus, and pertussis (whooping cough) as a five-dose series in infants and children 6 weeks to 7 years of age (prior to seventh birthday).
in allegato il bugiardino. A pagina 11 potete trovare
"Adverse events reported during post-approval use of Tripedia vaccine include idiopathic thrombocytopenic purpura, SIDS,
anaphylactic reaction, cellulitis, autism, convulsion/grand mal convulsion, encephalopathy, hypotonia, neuropathy, somnolence
and apnea. Events were included in this list because of the seriousness or frequency of reporting. Because these events are
reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequencies or to
establish a causal relationship to Components of Tripedia vaccine.2"
il vaccino è stato approvato negli USA dalla FDA






Inserted
Skipped
None
Removed